Context
Recently, the “Transmission Zero” project—a collaboration between Imperial College London and the Ifakara Health Institute in Tanzania—achieved a breakthrough by demonstrating that genetically modified mosquitoes could block malaria parasite transmission in a real-world endemic setting. This marks a shift from laboratory-only success to potential field application of Population Modification using CRISPR-Cas9 gene drives.
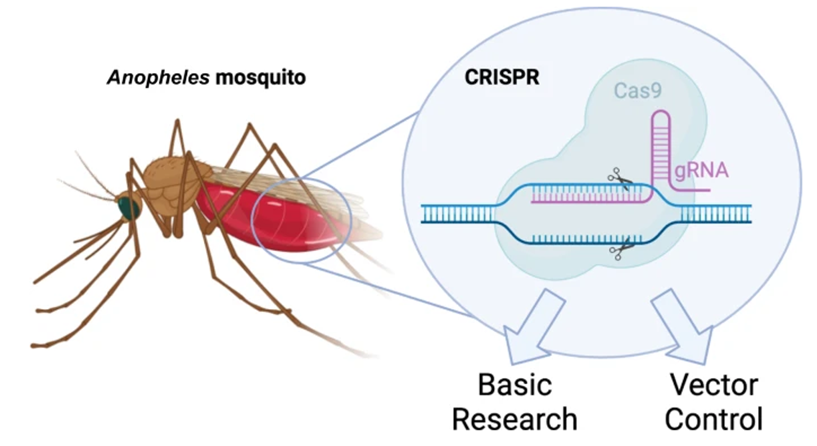
1. Understanding Gene Drive Technology
- Definition: A genetic engineering technology that bypasses traditional Mendelian inheritance to ensure a specific trait is passed to almost 100% of offspring (instead of the usual 50%).
- Mechanism: Uses the CRISPR-Cas9 system to “copy and paste” a modified gene into the partner chromosome during reproduction.
- Goal: Rapidly spread a desired trait through an entire wild population over a few generations.
2. Highlights of the Tanzania Study
- First for Africa: It demonstrated that modified mosquitoes could suppress malaria parasites from real-world infections, not just laboratory cultures.
- Localized Engineering: Conducted in a high-containment insectary in Bagamoyo, Tanzania, using local Anopheles gambiae mosquitoes.
- Effector Molecules: Engineered mosquitoes produced two antimicrobial peptides in their midgut upon taking a blood meal, preventing the Plasmodium parasite from reaching the salivary glands.
- Safety Features: Researchers are exploring “Self-limiting” drives (which disappear over time) and “Off-switches” to reverse the spread if needed.
3. Key Approaches in CRISPR-Cas9 Malaria Control
- Population Suppression (Gene Drives): CRISPR-Cas9 disrupts essential female reproduction genes (e.g., AGAP005958, AGAP011377), leading to a drastic reduction in mosquito numbers and, consequently, reduced transmission.
- Population Modification/Replacement: Mosquitoes are engineered to be resistant to the malaria parasite, making them unable to transmit it, such as through the knockout of the FREP1 gene.
- Precision-Guided Sterile Insect Technique (pgSIT): A specialized CRISPR approach that releases sterile males to reduce the population without using a self-sustaining gene drive.
4. Comparative Analysis: Malaria vs. Dengue
| Feature | Malaria | Dengue |
| Causative Agent | Protozoa (Plasmodium species like P. falciparum, P. vivax) | Virus (Flavivirus – DENV 1, 2, 3, 4) |
| Primary Vector | Anopheles Mosquito (Female) | Aedes aegypti (and Aedes albopictus) |
| Feeding Pattern | Mostly Night-biters (Dusk to Dawn) | Mostly Day-biters (Early morning/Late afternoon) |
| Breeding Habitat | Clean, stagnant water (Puddles, ponds, slow streams) | Artificial containers (Coolers, tires, pots, trays) |
| Incubation Period | Longer (Usually 10–15 days) | Shorter (Usually 3–14 days) |
| Key Symptoms | High fever with chills and rigors (shaking); cycling fever patterns. | High fever, severe “break-bone” joint/muscle pain, behind-the-eye pain, skin rash. |
| Major Complication | Cerebral Malaria, Severe Anemia. | Dengue Hemorrhagic Fever (DHF), Platelet drop. |
| Diagnostic Test | Blood Smear (Microscopy) or RDT (Rapid Diagnostic Test). | NS1 Antigen test (early), IgM/IgG Antibody tests. |
| Vaccine Status | RTS,S and R21/Matrix-M (Approved for children). | Dengvaxia (limited use); Qdenga. |
| Some Key Facts World Malaria Day, observed on 25th April annually, was established by the World Health Organization (WHO) in 2007 to raise awareness and drive action against malaria.Malaria is a life-threatening disease caused by the Plasmodium parasite, transmitted to humans by infected female Anopheles mosquitoes. Treatment & Prevention: Advanced with effective drugs like chloroquine and artemisinin, supported by tools like insecticide-treated nets (ITNs) and indoor spraying.Vaccine: In October 2021, the WHO recommended widespread use of the RTS,S/AS01 (Mosquirix) vaccine for children in sub-Saharan Africa and moderate-to-high malaria transmission areas.WHO E-2025 initiative: The World Health Organization (WHO) launched the E-2025 initiative in April 2021 to support 25 identified countries in eliminating malaria by 2025. |
Which one of the following statements best reflects the idea behind the Precision-Guided Sterile Insect Technique (pgSIT) often discussed in the context of malaria control?
(a)Genetically modified mosquitoes are released to directly kill malaria parasites inside the human body.
(b) Sterile male mosquitoes are released into the wild to mate with females, leading to no viable offspring.
(c) Mosquitoes are engineered to migrate away from human habitats permanently using genetic modification.
(d) Female mosquitoes are genetically modified to produce vaccines against malaria in their saliva.
Answer: B
Explanation:
• pgSIT uses CRISPR-based genetic engineering to produce sterile male mosquitoes.
• These males mate with wild females, but no offspring survive, reducing mosquito population over time.
• It is non–self-sustaining, unlike gene drive systems.