Context
- Recently a study reported the development of a new generation of immune cells with HIT (HLA-independent T-cell) receptors designed to overcome the “faint signal” problem in solid tumours such as kidney, ovarian, and pancreatic cancers.
- Unlike traditional CAR-T therapy—which has been highly effective in blood cancers but less so in solid tumours due to antigen heterogeneity—these receptors enable engineered T-cells to detect and target cancer cells that express very low levels of tumour markers.
What is CAR-T Cell Therapy?
Chimeric Antigen Receptor (CAR) T-cell therapy is a revolutionary form of immunotherapy that falls under the category of gene therapy. Unlike chemotherapy, which uses chemicals to kill cancer, CAR-T therapy uses the patient’s own immune system as a weapon.
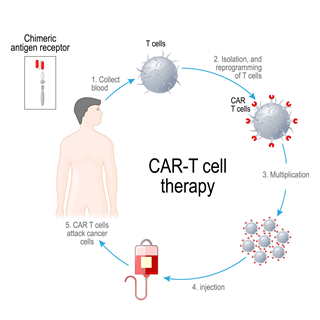
1. The Core Components
- T-Cells: These are a type of white blood cell (lymphocyte) responsible for identifying and killing foreign pathogens or abnormal cells.
- CAR (Chimeric Antigen Receptor): This is an artificial receptor engineered in a lab. It does not occur naturally. The term “chimeric” refers to the fact that it is composed of parts from different sources (an antibody part to see the cancer and a T-cell part to activate the attack).
2. The Process: From Patient to Cure
- Apheresis: T-cells are extracted from the patient’s blood.
- Genetic Engineering: In a laboratory, a viral vector is used to “recode” these T-cells. A new gene is inserted so the cells grow the CAR on their surface.
- Expansion: These newly engineered cells are multiplied into millions.
- Infusion: The “supercharged” T-cells are infused back into the patient.
- Targeting: The CAR acts like a GPS, allowing the T-cells to lock onto a specific protein (usually CD19) on cancer cells and destroy them.
NexCAR19: India’s Indigenous Pride
- Developed by: ImmunoACT (a company incubated at IIT Bombay) in collaboration with Tata Memorial Centre.
- Approval: It was approved by the Central Drugs Standard Control Organization (CDSCO) in October 2023.
- Significance: It is a “humanized” therapy, meaning it uses human-like protein sequences to reduce the risk of the body rejecting the treatment.
- Cost Factor: While global treatments cost approximately ₹3–4 crore, NexCAR19 has brought the cost down to ₹30–40 lakh, with targets to reach ₹10 lakh in 2026.
Key Advantages and Risks
| Feature | Description |
| “Living Drug” | Unlike pills that leave the body, these cells can persist for years, providing long-term surveillance against cancer recurrence. |
| Precision | It specifically targets cancer cells while sparing most healthy cells, unlike the “scorched earth” approach of chemotherapy. |
| Major Side Effect | Cytokine Release Syndrome (CRS) – a massive immune response that can cause high fever and organ failure if not managed. |
| Current Limit | Highly effective for blood cancers (Leukemia, Lymphoma) but struggles with solid tumors (like lung or breast cancer) due to their protective microenvironment. |
Q. With reference to CAR T-cell therapy, consider the following statements:
1. It involves the genetic modification of a patient’s own immune cells to recognize and attack cancer.
2. It is currently the primary standard of care for solid tumors like lung and liver cancers in India.
3. Unlike chemotherapy, CAR T-cells are considered a "living drug" as they can persist in the body for long periods.
Which of the statements given above is/are correct?
(a) 1 only
(b) 1 and 3 only
(c) 2 and 3 only
(d) 1, 2, and 3
Answer: B
Solution:
• STATEMENT 1 IS CORRECT: The therapy extracts T-cells and genetically modifies them with a Chimeric Antigen Receptor (CAR) to target cancer antigens.
• STATEMENT 2 IS INCORRECT: While research is ongoing for solid tumors, CAR T-cell therapy is currently approved and effective primarily for blood cancers (hematological malignancies) like Leukemia and Lymphoma. Solid tumors have a hostile environment that currently limits the therapy's efficacy.
• STATEMENT 3 IS CORRECT: They are called "living drugs" because the engineered cells multiply and can remain active in the patient's bloodstream for months or years to prevent relapse.